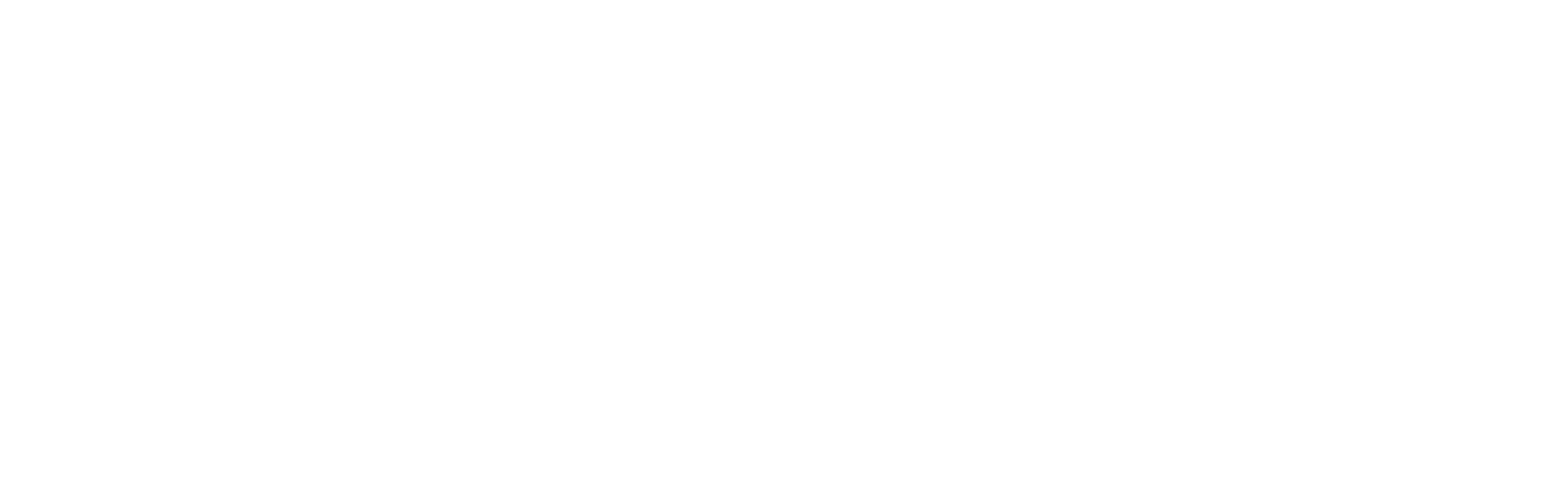
Youth triumphs in test to regenerate kidney tissue

Youth triumphs in test to regenerate kidney tissue
Stem cells implanted into mice produced improved kidney grafts
Scientists today announced the discovery that, in tests to develop multifunctional kidney tissue, young stem-cell populations showed dramatic advantages compared with organoids that were just one week older.
Results of their experiment were published in January in the journal The Innovation.
In the study, lab-grown mini-kidneys only 4 days old, after being implanted in mice, went on to develop specialized tissues not seen in previous attempts. By contrast, the same mini-kidneys that had been allowed to mature for two weeks generally struggled to develop in a mouse body.

“We’re starting with stem cells, and the main question we’re trying to answer is: What is the right stage to put the cells into a host? What's the window of opportunity to make the best grafts?” explained Benjamin Freedman.
He is the paper’s senior author and an associate professor of nephrology at the University of Washington School of Medicine.
What are organoids’ prospective value?
Over the past decade, Freedman’s kidney organoid systems have become reliable models for researching disease mechanisms and for exploring tissue generation. These organ-like structures derive from human pluripotent stem cells, which can be developed into any bodily tissue.
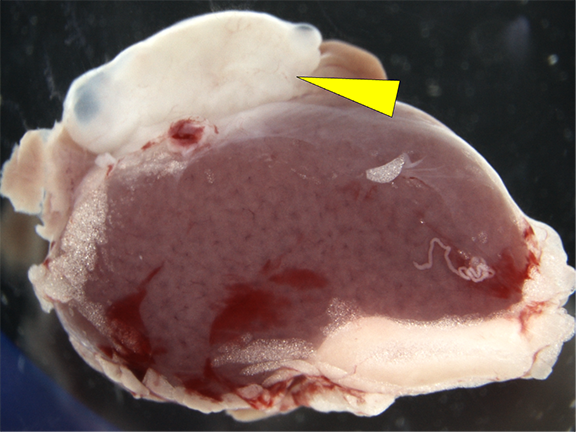
One problem Freedman and others hope to address, ultimately, is the persistent, deep deficit of available donor organs. Upward of 100,000 people are on the national kidney transplant waiting list and around 14% of American adults have chronic kidney disease — a trend that’s worsening.
Researchers in this field first must identify the strategy that holds the most promise for tissue generation, he said.
“The results so far support the idea of essentially injecting stem cells into a body and having them take form and make connections to create kidney tissues in the body,” Freedman said.
There, stem cells have access to blood vessels that provide greater nourishment than what they would receive outside of the body. This gives the cells the opportunity to develop “chimeric” tissue that is part host and part donor.
Younger cells show vigor, adaptability
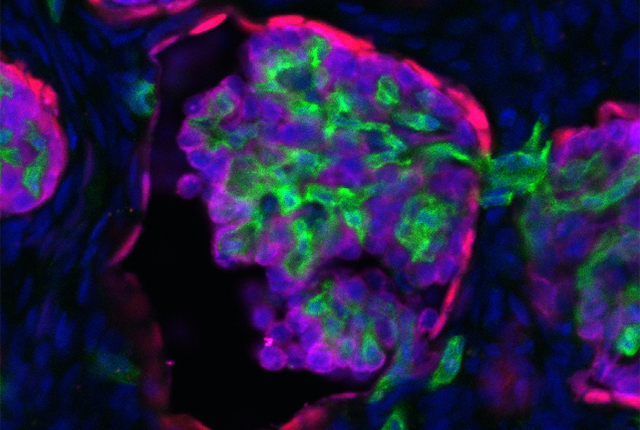
One of the keys to success was the presence of stromal progenitor cells within the younger cell cultures. These stromal progenitors gave rise to mesangial cells, a specialized population that gives structure to kidney tissues.
In previous implant studies, Freedman had never seen mesangial cells in the end-result specimens. This time, they were there, but only in grafts originating from the younger cell population.
“It's all about timing. We started with the pluripotent stem cell, which we gave a little direction to turn into a kidney. The stromal progenitor cell never survives very long outside of the body. So we shortened the window to implant, and the stromal cell descendants were able to survive and change into mesangial cells.”
By comparison, in the cell populations that had been allowed to mature for another week as organoids, stromal cells were absent. As implants, the older organoid cells were prone to develop cysts and lose their structure, Freedman noted.
Freedman said that the result might be attributed to the younger cells' greater potential, compared to older cells that were more set in their ways.
Next steps
Given the kidney’s key role to eliminate waste, getting organoid progenitor cells to connect with a host’s urinary-system cells “is the piece we’ll have to tackle next,” Freedman added. This will require the researchers to discover a more apt implant site.
“We've been implanting cells in a little pocket next to the kidney called the capsule, but we aren’t seeing that [the cells] form functional connections with the rest of the kidney from that site. We need to find a better spot.”
The paper’s first author was Thomas Vincent, a UW graduate student in Bioengineering who works in Freedman’s lab. Other co-authors included Samera Nademi, a postdoctoral fellow in the Freedman lab, and collaborators at the Sheba Medical Center in Tel Aviv, Israel.
The work was supported by Cystinosis Research Foundation (CRFF-2021-003, CRFF-547-2023-004), National Institutes of Health (UC2DK126006), a Regular Award from the United States-Israel Binational Science Foundation, and an Institute for Stem Cell and Regenerative Medicine Fellows award.